Bisphenol A Suppliers, Dealers in Mumbai, Maharashtra, India
Bisphenol A
Bisphenol A (BPA) is a high volume industrial chemical. It is widely used in making polycarbonate plastics as well as epoxy resins. With its aromatic backbone & hydroxyl moieties, BPA serves as a critical building block. It lends strength & durability to numerous everyday products—from water bottles as well as food-storage containers to coatings lining metal packaging. However, its ubiquity also triggers safety concerns, particularly related to hormonal activity in the body. This detailed profile explores BPA’s chemistry, applications, specifications as well as safe handling, while highlighting why Shakti Chemicals is a trusty provider.
What is Bisphenol A
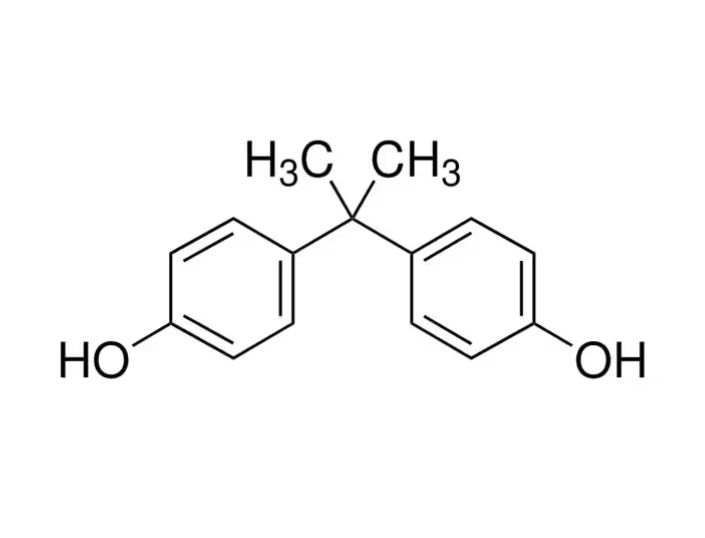
Bisphenol A (IUPAC: 4,4' isopropylidenediphenol; CAS NO. 80 05 7) is a difunctional phenolic compound produced via the acid-catalyzed condensation of acetone with phenol. The result is a crystalline, slightly soluble white solid (≈0.3 g/L at 25 °C) with a melting point near 155–156 °C, density ~1.217 g/cm³, and negligible vapour pressure. The molecule features two hydroxyphenyl groups joined by an isopropylidene bridge. It serves as a adamant & reactive core in polymer synthesis. Factory wise, 65–70% of BPA is used to make polycarbonate (PC) plastics. It is a priced product. It is valued for clarity, toughness & dimensional stability, while 25–30% is committed to epoxy & vinyl ester resins. Additional niche uses include antioxidants (e.g., in brake fluids), thermal-paper developers as well as minor additives in PVC and polyurethanes. Although BPA is not a plasticizer, its phenolic backbone is indispensable in forming long polymer chains. Global production exceeded ~10 million tonnes in 2022. Despite wide utility, BPA is a known endocrine disruptor (xenoestrogen), prompting regulatory scrutiny, especially in food-contact materials and baby products.
Chemical Specifications
| Property | Details |
|---|---|
| CAS Number | 80-05-7 |
| Chemical Name | Bisphenol A (4,4'-isopropylidenediphenol) |
| Molecular Formula | C15H16O2 |
| Appearance | White crystalline solid |
| Melting Point | 155–156 °C |
| Molecular Weight | 228.29 g/mol |
| Density | ~1.217 g/cm³ |
| Solubility in Water | 0.3 g/L at 25 °C |
| LogP (octanol–water) | ~3.41 (hydrophobic) |
| Vapour Pressure (25 °C) | Negligible |
| Flash Point | ≈227 °C |
| NFPA Ratings | Health 2, Flammability 1, Reactivity 0 |
Technical Specifications
Polycarbonate-grade Bisphenol A (e.g., BPA-157)
Purity: typically ≥99%
Color (Pt-Co): <200
Free Phenol: <0.05 % w/w
Freezing Point: ≥156.5 °C
Iron Content: <0.5 mg/kg
Bulk Density: ~560–650 kg/m³ (35–41 lb/ft³)
Form: white flakes/solid
Structure of Bisphenol A
BPA features two phenolic rings connected via a methylidene bridge (-C(CH₃)₂-). The rings often adopt a torsion angle near 91.5°. It contributes to crystalline packing. The hydroxyl groups at the para positions are pivotal reactive sites enabling condensation with phosgene (to form PC) or epichlorohydrin (for epoxy resins).
Here is a basic line-structure formula of BPA:
C15H16O2
Or in SMILES notation:
CC(C1=CC=C(C=C1)O)C(C2=CC=C(C=C2)O)
Each C6H4 represents a benzene ring with a hydroxyl (-OH) group at the para position.
These two rings are joined via a central carbon attached to two methyl groups (–CH₃). It forms an isopropylidene bridge.
Molecular Formula: C₁₅H₁₆O₂
IUPAC Name: 4,4′-(propane-2,2-diyl)diphenol
Handling and Storage
Referencing typical SDS recommendations:
Handling: Avoid dust generation; use local exhaust ventilation. Ground/bond all equipment to prevent static discharge. Wear protective gear—gloves, glares, lab coat.
Storage: Store in cool (<50 °C), arid as well as well-ventilated areas. Store it away from strong oxidizers, acids, or bases. Containers should be tightly packed or vacuumed. Nitrogen blanketing is advisable for bulk storage.
Spills: Sweep/collect material without creating dust. Dispose this as per regulations. Vacuuming with explosion-proof products recommended.
Fire Hazard: Combustible dust—minimize ignition sources. Use water spray, CO₂, or dry chemical extinguishers.
Reactivity: Stable under normal conditions; reactive with oxidizing agents. No risky or hazardous polymerization.
Why Choose Shakti Chemicals
As a top rated global supplier, exporter, trader, dealer, exporter, importer or distributor of specialty chemicals or acids, Shakti Chemicals in Mumbai, India guarantees best-in-class quality as well as service:
High-Purity Grades: Industrial and technical-grade BPA, meeting stringent global specs.
Comprehensive Services: As dedicated suppliers and distributors in Maharashtra, they manage sourcing, logistics, documentation as well as delivery end-to-end.
Tailored Packaging & Support: Customizable pack sizes, full technical support, and prompt SDS/MSDS delivery.
Global Compliance: Full regulatory adherence for export markets, with secure packaging and cold-chain options.
User-Centric Operations: Blending supplier reliability with athletic distribution, backed by transparent pricing, JIT supply & also reliable warehousing.
Product Pack Size
Shakti Chemicals typically offers:
Drums: 200–250 kg
IBC Totes: ~1,000 kg
Bulk Bags: 500–1,000 kg
Hopper Trucks or Railcars: Ideal for large-scale industrial users
Custom Packaging: Upon request, tailored to customer requirements.

Applications of Bisphenol A
Polycarbonate Plastics – It is transparent, impact-defiant items (optical lenses, medical & food packaging, CDs/DVDs).
Epoxy Resins – Interior coatings for food cans, industrial/residential paints, adhesives & pipe linings.
Vinyl Ester Resins – Corrosion-defiant composites for marine/industrial applications.
Antioxidants – Additive in brake fluids as well as hydraulic systems.
Thermal Paper Developers – Reactant in thermal print media (e.g., receipts).
Specialty Polymer Additives – Minor use in phenolic resins, polyurethanes, PVC.
Analytical Standards – Certified reference material for research/quality labs.
FAQs
What is Bisphenol A used for?
It is widely used to manufacture polycarbonate plastics (~65%) as well as epoxy resins (~25–30%).
What is the chemical formula as well as mol. weight?
The chemical formula is C₁₅H₁₆O₂ & Molecular weight is Mw ≈228.29 g/mol.
Is BPA soluble in water?
Poorly—≈0.3 g/L at 25 °C; more soluble in organic solvents.
What health risks does BPA pose?
It’s a endocrine disruptor; EFSA recently cut tolerable daily intake to 0.2 ng/kg/day, citing consumer exposure concerns.
How should BPA be stored safely?
In sealed containers, ≤50 °C. Put away from fire catching sources, strong oxidizers. Also nitrogen blanketing recommended.
How to handle BPA dust safely?
Try to decrease dust by using grounded & ventilated equipment. Always wear PPE protection & clean spills without air-blowing.
Can BPA catch fire?
Solid BPA is combustible as dust. It's flash point is ≈227 °C. Try to extinguish with water spray, CO₂ or dry chemical.
What grades are available?
Industrial/technical grades (≥99%) for PC as well as epoxy applications. Special flake-grade like BPA 157 meets PC quality specs.
How is BPA transported?
It is transported in drums, IBCs, and bulk bags. Coastal rail/truck shipments need grounding as well as inerting. It’s not UN regulated as bulk solid but demands safe handling.
Why purchase from Shakti Chemicals?
Offers genuine quality, global adherence, tailored packaging & also expert user-oriented services across the supply chain.
Bisphenol A remains a cornerstone chemical in modern materials science, enabling the manufacture of rigid polycarbonates and durable epoxy/vinyl ester resins. While its efficacy is unquestioned, evolving health assessments have started stricter controls as well as safer handling. Shakti Chemicals stands out as a reliable international supplier, trader, dealer, importer or distributor. It offers high purity BPA, comprehensive technical support, compliant logistics & also user-focused answers. Whether for large-scale industrial use or specialized applications, seeking products from Shakti Chemicals guarantees uniform quality, dependable supply as well as regulatory excellence.
Get in Touch with Us
Looking for high-quality Bisphenol A? Contact us today for bulk orders and technical details.
Contact Us- Address: 206-207, 2nd Floor, Wadala Udhyog Bhavan, Naigaon Cross Road, Wadala (West)
- Email: info@shaktichemicals.org
- Phone: +91 4044 0000
