Methyl Sulfonyl Chloride Suppliers, Dealers in Mumbai, India
Methyl Sulfonyl Chloride – A Key Intermediate in Chemical Synthesis
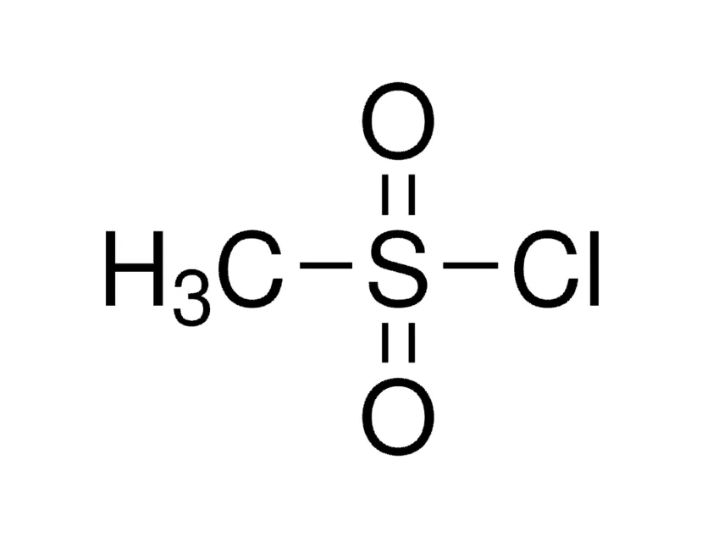
Methyl Sulfonyl Chloride or MSC is a significant organosulfur compound. This is extensively utilized across chemical synthesis industries. With the formula CH₃SO₂Cl, it offers both versatility as well as reactivity. It serves as a key building block in pharmaceuticals, agrochemicals, dyes, polymers & also specialty intermediates. Its high electrophilicity at the sulfur center makes it ideal for introducing sulfonyl chloride functionality into diverse organic molecules. This allows further substitution reactions. In addition, its relatively facile handling under controlled conditions. This thing has made MSC a preferred reagent in laboratory as well as industrial environments.
What is Methyl Sulfonyl Chloride?
Methyl Sulfonyl Chloride is chemically shown as CH₃SO₂Cl. It is a sulfonyl chloride derivative carrying a methyl group attached to a sulfonyl moiety. This thing is further bonded to a chlorine atom. Its role as a reactive intermediate stems from the great polarized S–Cl bond, which facilitates nucleophilic attack. This makes it an excellent choice for sulfonation, sulfonamide formation as well as installation of sulfonyl functionalities in complex molecules.
Physically, MSC is a colorless to pale yellow liquid under ambient conditions, with a pungent odor and moderate vapour pressure. It is peculiarly made by the chlorination of methyl sulfonic acid or oxidation of methylthio compounds. This reaction is then followed by chlorination. Its importance extends to pharmaceuticals. Specifically in the synthesis of sulfonamide antibiotics as well as antiviral agents. Here the sulfonyl chloride moiety is a key mediator. In agrochemical R&D, MSC is used to introduce sulfonyl-based bioactive groups, this changes molecular stability & also efficacy.
Beyond pharmaceuticals and agrochemicals, MSC aids in generating specialty dyes, where it enables sulfonylation of aromatic intermediates and enhances dye molecule solubility. It is also used in polymer modification as well as new-age materials. This serves as a cross-linker or functionalizing agent. The chemical's utility is underpinned by its selectivity. It reacts preferentially with amines, alcohols & also phenols to form sulfonamides, sulfamates as well as sulfonate esters under controlled conditions. Overall, Methyl Sulfonyl Chloride is a valuable and widely used sulfonylating agent in modern organic synthesis.
Product Description Table
| Property | Details |
|---|---|
| CAS Number | 124-63-0 |
| Chemical Name | Methyl Sulfonyl Chloride |
| Molecular Formula | CH3SO2Cl |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent, irritating |
| Molecular Weight | 114.55 g/mol |
| Purity | ≥ 99 % (industrial grade and high-purity lab grades) |
| Assay Method | GC / HPLC |
| Boiling Point | 145–147 °C (at 760 mmHg) |
| Density | 1.445 g/cm³ at 20 °C |
| Refractive Index | n₂₀ᴅ = 1.495–1.505 |
| Flash Point | 75 °C (closed cup) |
| Solubility | Reacts with water; soluble in chlorinated and polar aprotic solvents like DCM, THF |
| Storage Condition | ≤ 25 °C, dry and well-ventilated area |
| Expiration | 12 months from date of manufacture only if stored properly |
| Packaging | HDPE-lined drums / aluminum bottles / stainless steel containers (customized) |
Specifications
1. Assay: ≥ 99 %
2. Density (20 °C): 1.445 ± 0.005 g/cm³
3. Boiling Point: This is between 145–147 °C at 760 mmHg
4. Moisture Content: ≤ 0.05 % (Karl Fischer titration)
5. Assay Method: GC or HPLC analysis
6. Chloride as HCl: ≤ 0.10 %
7. Heavy Metals: This need to be ≤ 10 ppm
8. Acidic Impurities: ≤ 0.05 %
9. Storage Stability: No significant degradation over 12 months when stored as per rules

Handling and Storage
Storage Conditions
Store MSC in tightly sealed, moisture proof containers (HDPE, aluminum, or stainless steel). Keep the material in a cool (≤ 25 °C), non-humid as well as ventilated chemical storage area away from incompatible substances.
Incompatibilities
Avoid contact with water, alcohols, amines, bases, oxidizers & also strong reducing agents. Reaction with water in the end gives corrosive HCl gas.
Handling Precautions
– Always handle under a chemical fume hood or in a well ventilated safe enclosure.
– Use personal protective equipment: chemical resistant gloves (e.g., nitrile), goggles, face shield, lab coat, and where high volume use is involved, chemical resistant apron.
– Ensure immediate access to eyewash stations and safety showers.
– Transfer using closed systems or syringes equipped with PTFE plugs to minimize atmospheric exposure.
Disposal
– Neutralize residual MSC with mild base (e.g., 5 % sodium bicarbonate in a fume hood).
– Follow all local, state, and national hazardous waste disposal regulations.
– Provide documentation when shipping waste to appropriate disposal facilities.
Structure of Methyl Sulfonyl Chloride
Chemically expressed as CH₃–S(=O)₂–Cl, MSC is a sulfonyl chloride. The central sulfur atom is tetrahedrally coordinated to: one methyl group, two double-bonded oxygen atoms, and one chlorine atom. The presence of two strongly electronegative oxygens and chlorine atoms generates a polarized S–Cl bond, making sulfur highly susceptible to nucleophilic attacks.
Bond geometry: Sulfur adopts a distorted tetrahedral geometry with approximate bond angles of 107–109° due to lone pairs and bonding domains.
Electron distribution: The S=O bonds create dipoles, and the sulfur–chlorine bond is strongly polarized (δ+ on S, δ– on Cl), enabling the chlorine to act as a good leaving group.
Reactivity: The polarized S–Cl bond readily undergoes substitution with nucleophiles (e.g., amines, alcohols, phenols), enabling formation of sulfonamide, sulfamate, or sulfonate products. Mild reaction conditions are often sufficient, making MSC a selective reagent.
Why Choose Shakti Chemicals
Choosing a reliable vendor for Methyl Sulfonyl Chloride is crucial for consistent quality and regulatory compliance. Shakti Chemicals stands out due to:
Experienced Suppliers, exporter: Decades of expertise in producing high-purity organosulfur compounds using robust GMP-compliant facilities.
Global Traders: Serving worldwide customers with custom packaging, documentation, and logistics support.
Industrial Dealers: Stocking ready inventory of MSC to ensure reduced lead times and prompt dispatch.
Specialized Importers: Navigating customs, export–import regulatory frameworks to ensure seamless international delivery.
Dedicated Distributors: Establishing local warehousing and technical support networks for immediate availability.
Key differentiators include:Quality Assurance:
– High-purity MSC grades (≥ 99 %), tested using GC/HPLC.
– Full analytical documentation with COA, MSDS, stability data.
Custom Offerings:
– Packaging from 1 kg lab-scale bottles to bulk tonnes.
– Customized labeling, dual certification, and specialty packaging to meet GLP/GMP requirements.
Regulatory Compliance:
– RoHS, REACH, TSCA regulations covered with full traceability and documentation.
Technical & Customer Support:
– Product usage guides, reagent selection assistance, and on-demand safety training.
– Tailored consultation for scale-up synthesis or complex process development.
Reliable Logistics:
– Cold-chain options for stability-critical payloads; insurance and regulatory paperwork provided end-to-end.
Whether you’re ordering a few grams or tons, Shakti Chemicals offers stable supply chains and expert support as trusted suppliers, traders, dealers, importers, and distributors.
Product Pack Size
Shakti Chemicals offers Methyl Sulfonyl Chloride in a variety of packaging options tailored to customer needs:Laboratory-scale:
– 100 mL, 250 mL glass bottle (HDPE-lined cap)
– 500 mL / 1 L amber glass with PTFE-lined screw cap
Pilot plant / R&D:
– 5 kg / 10 kg sealed canisters
– 25 kg HDPE-lined steel pails
Industrial / bulk:
– Drums: 50 kg and 200 kg stainless steel or HDPE-lined steel drums
– ISO-tank and IBC options for >500 kg orders
Customization:
– Nitrogen-blanketed packaging
– UN-rated containers, export-grade wooden crates
– Custom labeling, bar-coding, batch tracking
Applications of Methyl Sulfonyl Chloride
Pharmaceutical Synthesis
Key intermediate for sulfonamides, sulfamates, and sulfone pharmaceuticals (antimicrobials, antivirals).
Useful in medicinal chemistry for introducing sulfonyl groups to enhance pharmacokinetic and solubility profiles.
Agrochemical Manufacturing
Building block in herbicides and pesticides, enabling incorporation of sulfonyl functionalities for potency and environmental stability.
Specialty Dyes & Pigments
Sulfonation of aromatic intermediates enhances water solubility & dye uptake in textile applications.
Polymer & Material Chemistry
Used to functionalize polymers or as a cross-linker, introducing sulfonate groups for ion-exchange, enhanced hydrophilicity, or thermal stability.
Fine Chemicals & Intermediates
In specialty organic synthesis for pharmaceuticals, fragrances, and fine chemical intermediates requiring selective sulfonylation.
Analytical & Reagent Manufacture
Producing fluorescent probes, labeling reagents, and sulfonyl-activated tagging molecules for biochemical assays.
FAQs
1. Can Methyl Sulfonyl Chloride be shipped via air freight?
Yes—it’s classified as UN 2585 (Toxic liquid, organic, n.o.s.), Packing Group III. Air transport requires hazard labeling, safety datasheets, and trained handlers.
2. What is the reactivity with water?
Highly reactive—hydrolyzes to methylsulfonic acid and HCl. Strictly eliminate moisture exposure during storage and handling.
3. How do I detect moisture in MSC batches?
Karl Fischer titration is the preferred method, with moisture acceptance typically ≤0.05 %.
4. Does it require inert gas blanketing?
Recommended for large-scale or long-term storage. Nitrogen blanketing prevents moisture uptake and oxidative impurities.
5. Are there specific analytical methods for purity?
Yes—Gas Chromatography (GC) or High-Performance Liquid Chromatography (HPLC) calibrated with certified standards.
6. What solvents are recommended for reaction?
Aprotic solvents like dichloromethane (DCM), chloroform, and tetrahydrofuran (THF). Reactions in polar protic solvents risk HCl evolution as well as competitive substitution.
7. Can MSC be used directly in amine–sulfonylation?
Absolutely. It reacts readily with primary and secondary amines to form sulfonamides, often conducted at 0–25 °C with bases like triethylamine or pyridine.
8. What PPE is essential?
Eye protection (goggles/face shield), chemical-resistant gloves (nitrile or neoprene), lab coat/apron, and fume hood use are mandatory.
9. What are typical reaction yields?
Yields of 80–95 % are common for sulfonamide formation under optimized conditions; industrial processes often exceed 90 % after purification.
10. Is MSC approved under REACH/TSCA?
Yes—Shakti Chemicals ensures its MSC is fully registered and compliant with REACH, TSCA, and other international regulatory frameworks.
Methyl Sulfonyl Chloride is an all-rounder as well as essential reagent in modern chemical synthesis. It is prized for its efficiency in introducing sulfonyl functionality into complex molecules. From pharmaceuticals and agrochemicals to dyes and advanced materials, MSC offers precise reactivity and compatibility with various nucleophiles. When sourced from a reliable partner like Shakti Chemicals in Maharashtra—expert suppliers, dealers, traders, importers, and distributors —users benefit from superior quality, full documentation, customized packaging, and technical support. With its strong manufacturing protocols as well as global compliance capabilities, Shakti Chemicals in Mumbai, India ensures that MSC usage remains safe, effective, and scalable. Whether for laboratory research or industrial-scale production, MSC is a high-impact reagent that delivers consistent results.
Get in Touch with Us
Looking for high-quality Methyl Sulfonyl Chloride? Contact us today for bulk orders and technical details.
Contact Us- Address: 206-207, 2nd Floor, Wadala Udhyog Bhavan, Naigaon Cross Road, Wadala (West)
- Email: info@shaktichemicals.org
- Phone: +91 4044 0000
