Di-Isopropyl Ether Suppliers, Dealers in Mumbai, India
Di-Isopropyl Ether – High-Purity Solvent
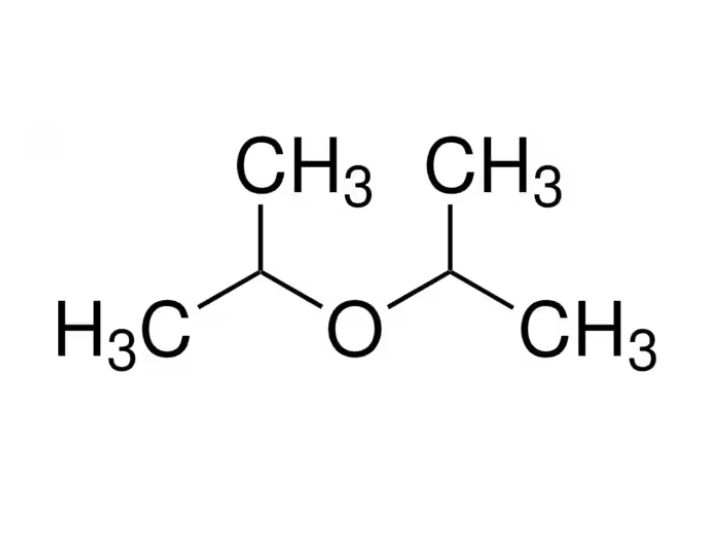
Di-Isopropyl Ether (DIPE) is a versatile organic compound. It is widely used in the chemical industry as a solvent as well as in extraction processes. It is renowned for its high volatility as well as less water solubility. This finds applications in both laboratory & industrial settings. As an ether derivative, it possesses strong flammability & must be handled with care. Specifically in large-scale usage. Its unique chemical structure allows it to efficiently separate or extract organic substances, especially polar as well as non-polar compounds. Due to these properties, Di-Isopropyl Ether has earned a stable position in various sectors like pharmaceuticals, petrochemicals, coatings, and synthetic chemistry.
What is Di-Isopropyl Ether?
Di-Isopropyl Ether (DIPE), chemically named as 2-isopropoxypropane, is a secondary ether synthesized by the dehydration of isopropyl alcohol under acidic conditions. It is a colorless & highly flammable liquid with a characteristic ether-like odor. DIPE’s formula is (CH₃)₂CHOCH(CH₃)₂, & it belongs to the class of dialkyl ethers. It has a relatively low boiling point as well as is less dense than water. Due to its hydrophobic nature & immiscibility with water, DIPE serves as a prominent solvent for organic extractions.This is particularly beneficial in refining & purification of pharmaceuticals as well as fine chemicals.
One of the greatest features of this compound is its ability to dissolve both polar as well as non-polar compounds. This makes it a valuable solvent in complex reaction systems. However, due to the possibility of peroxide formation upon prolonged exposure to air & also light, DIPE needs proper stabilizers & also careful storage. It’s extensively used in the separation of organic acids & in azeotropic distillation processes. Here it aids in remove water from chemical reactions.
In laboratories, DIPE is often used as an extraction agent to purify reaction mixtures. In industrial contexts, it plays a role in fuel production, especially in high-octane gasoline formulations. Its solvency properties also make it ideal for cleaning, degreasing as well as surface preparation tasks.
Product Description Table
| Property | Details |
|---|---|
| CAS Number | 108-20-3 |
| Chemical Formula | (CH₃)₂CHOCH(CH₃)₂ |
| Molecular Formula | C6H14O |
| Odor | Ether-like |
| Boiling Point | > 68–70°C |
| Molecular Weight | 102.18 g/mol |
| Melting Point | -60°C |
| Density | Di isopropl ether |
| Checmical Name | 0.725 g/cm³ at 20°C |
| Solubility in Water | Insoluble in waterc(0.88 g/L at 25°C) |
| Flash Point | -28°C (Closed cup) |
| Autoignition Temperature | 415°C |
| Vapor Pressure | 132 mmHg at 20°C |
Specifications
Purity: It is minimum 99.0%
Moisture Measure: It is maximum 0.05%
Peroxide Level: It is < 0.005%
Acidity (as Acetic Acid): It is maximum 0.002%
Residue on Evaporation: It is maximum 0.005%
Refractive Index (n20/D): 1.375 - 1.377
Color (APHA): Max 10
Specific Gravity (25°C): 0.720–0.730
These specifications guarantee high quality, specifically when the DIPE is used in sensitive applications such as pharmaceuticals as well as electronics-grade processes.

Handling and Storage
Di-Isopropyl Ether is highly flammable & also must be handled with rigorous safety measures:
Handling Guidelines:
Always use in a well-airy area or under a fume hood.
Wear chemical-defiant gloves, shades & flame hesistant lab coats.
Avoid direct contact with skin or eye & inhalation of vapors.
Storage Guidelines:
Store in tightly closed, amber-colored drums away from direct sunlight & ignition sources.
Keep away from oxidizing agents along with strong acids.
Add suitable peroxide inhibitors like BHT if storage extends beyond a few months.
Use grounded jars to prevent static discharge during transfer of substance.
Regular inspection for peroxide buildup is pivotal to avoid hazardous decomposition. Crums or jars should be clearly labeled & also dated upon opening.
Structure of Di-Isopropyl Ether
The chemical structure of Di-Isopropyl Ether is based on an ether linkage between two isopropyl groups. Its structural formula is:
CH₃–CH(CH₃)–O–CH(CH₃)–CH₃
This symmetrical molecular structure contributes to its low polarity and makes it a non-hydrogen bonding solvent. The oxygen atom bridges two bulky isopropyl groups, which affects its boiling point and steric interactions during chemical reactions.
Why Choose Shakti Chemicals?
When it comes to buying high-quality Di-Isopropyl Ether, Shakti Chemicals stands as a trusty name in the chemical distribution industry in Mumbai, Maharashtra. As one of the leading Di-Isopropyl Ether suppliers, traders, dealers, importers, exporters and distributors in India, we offer:
- Premium Quality Assurance: Our DIPE is rigorously tested and complies with industry-grade specifications to ensure reliability across industrial applications.
- Competitive Pricing: We believe in providing cost-effective answers without compromising on great quality.
- Widespread Distribution Network: We have a well-established logistics chain to ensure timely and secure delivery of bulk and small orders across the globe.
- Customer-Centric Service: Technical support, product customization, and 24/7 customer service ensure that your unique needs are always met.
- Regulatory Compliance: We meet all national as well as international safety & environmental regulations.
Choose Shakti Chemicals for reliable and consistent quality, whether you are a manufacturer, lab technician, or chemical processor.
Product Pack Size
Shakti Chemicals gives Di-Isopropyl Ether in multiple packaging options suited for laboratory, pilot plant & also industrial use:
500 ml Glass Bottles – For laboratory and research purposes
5 L HDPE Canisters – Ideal for pilot projects and testing labs
25 L Carboys – Common for medium-scale industrial use
200 L Drums – Suitable for large-scale applications
IBC Totes (1000 L) – Bulk storage and transportation
Customized packaging and labeling can also be arranged as per client requirements to maintain regulatory compliance and ease of use.
Applications of Di-Isopropyl Ether
- Pharmaceutical Industry:Used as a reaction solvent and in purification steps to isolate active pharmaceutical ingredients (APIs).
- Petrochemical Industry: Serves as a blending agent in high-octane fuels & also as a process solvent in refining.
- Paints and Coatings:Effective in dissolving resins as well as used in formulating surface coatings.
- Azeotropic Distillation: Employed to remove water from reaction mixtures during synthesis.
- Extraction Solvent:Helps in extracting organic acids and alkaloids from aqueous mixtures.
- Adhesive Formulations:Used to thin down glues and adhesives for industrial and consumer products.
- Laboratory Reagent:It’s preferred in analytical chemistry for liquid to liquid extraction techniques.
- Cleaning Agents:It is used in degreasing as well as cleaning precision instruments due to its quick evaporation rate.
- Agrochemical Processing:/bA solvent medium in the formulation of pesticide and herbicide concentrates.
- Perfume and Fragrance Industry:Occasionally used as a volatile carrier solvent for fragrance components
FAQs
Is Di-Isopropyl Ether safe to use?
It is safe under controlled conditions, but due to flammability and peroxide formation, appropriate precautions must be taken.
What industries use DIPE most?
Major consumers include the pharmaceutical, petrochemical, paint, and agrochemical industries.
Can DIPE form explosive peroxides?
Yes. Prolonged exposure to air and light can lead to peroxide formation; hence stabilizers are necessary.
How is DIPE different from diethyl ether?
DIPE has higher steric hindrance and a slightly higher boiling point compared to diethyl ether, making it suitable for specific reactions.
Is Di-Isopropyl Ether miscible with water?
No. DIPE is practically immiscible with water, which makes it effective for aqueous extraction.
Can I use DIPE as a fuel additive?
Yes. In some high-performance fuel applications, DIPE acts as an oxygenate additive.
How should DIPE be disposed of?
It should be incinerated in a chemical incinerator equipped with an afterburner and scrubber, in compliance with local regulations.
What is the shelf life of DIPE?
Typically 6–12 months if stored properly with stabilizers and away from light and air.
Does DIPE have any environmental impact?
Being volatile, it contributes to smog formation if released into the air. It should be contained and recycled or disposed of properly.
Is DIPE suitable for chromatography?
It can be used in non-polar chromatographic separations, although not as commonly as other solvents.
Di-Isopropyl Ether is a crucial chemical with diverse applications across industries. Its high purity, excellent solvency, and low water solubility make it ideal for processes requiring effective separation and purification. However, due care must be taken in its handling due to its flammability and peroxide risk. With Shakti Chemicals as your dedicated Di-Isopropyl Ether supplier, trader, importer, and distributor, you receive not only top-grade chemicals but also dedicated support and reliable delivery systems. Whether you're in pharmaceuticals, petrochemicals, or advanced material synthesis, DIPE can be a valuable addition to your chemical toolkit.
Get in Touch with Us
Looking for high-quality Di-Isopropyl Ether? Contact us today for bulk orders and technical details.
Contact Us- Address: 206-207, 2nd Floor, Wadala Udhyog Bhavan, Naigaon Cross Road, Wadala (West)
- Email: info@shaktichemicals.org
- Phone: +91 4044 0000
